Blog
How would you describe what "science" is?
Science is the act of curiosity. We discover through science. Science is discovery, and history, and math, and zoology. Science is the only reason we have information.
Update:
Science is observation and experimentation. It is finding truth. Nothing is real unless science finds proof to show that it is real. Science provides practical reasoning. It deciphers real life from myth.
what is a hypothesis, and why would you make one?
A hypothesis is a guess, it is what you predict the outcome of your experiment will be. We make hypothesis' because science can change our thinking. What you assume or what seems obvious may not be true when you test it.
Science is the act of curiosity. We discover through science. Science is discovery, and history, and math, and zoology. Science is the only reason we have information.
Update:
Science is observation and experimentation. It is finding truth. Nothing is real unless science finds proof to show that it is real. Science provides practical reasoning. It deciphers real life from myth.
what is a hypothesis, and why would you make one?
A hypothesis is a guess, it is what you predict the outcome of your experiment will be. We make hypothesis' because science can change our thinking. What you assume or what seems obvious may not be true when you test it.
Combustion
Annotations on Combustion Science:
(Some examples from my annotations)
What is fire safety in space?
What are microgravity conditions?
What does buoyancy-induced flows mean?
What does sedimentation mean?
Annotations:
(Some examples from my annotations)
What is fire safety in space?
What are microgravity conditions?
What does buoyancy-induced flows mean?
What does sedimentation mean?
Annotations:
Burning Balloon/Combustion
What happened when we burned our balloons? Why did this happen?What do you think happened when we burned our hydrogen balloons?
It seemed like the balloon exploded. The helium that we created from the citric acid, magnesium, and water had a reaction to the fire. First, it burned through the balloon, then the helium seemed like it caught fire as well. I think that the explosive noise was due to the balloon popping. The fire being stretched out was because of the helium atoms jumping around and releasing into all different directions.
UPDATE:
Oxygen got it's placement in the Periodic Table of the Elements because of its reactivity. This was how the Periodic Table of the Elements was made a long time ago. Electrons rule an elements reactivity. Oxygen has eight protons and eight electrons. It is on the hunt for two more electrons to fill the outer shell and make a stable atom.
There are three oxygen atoms bound to a central nitrogen atom. There is an initial spark of heat that breaks these bonds causing the oxygen to rush away from the Nitrogen to bond with the atoms that it prefers like carbon, hydrogen and even other oxygen atoms leaving the nitrogen to pair up with each other. Every time atoms form a new bond, the reaction releases a new energy which causes the explosion.
Another example of this is in a candle. The carbon atoms in the wick bond with the oxygen atom in the air. Or a very slow process of rust: when iron and oxygen combine.
It seemed like the balloon exploded. The helium that we created from the citric acid, magnesium, and water had a reaction to the fire. First, it burned through the balloon, then the helium seemed like it caught fire as well. I think that the explosive noise was due to the balloon popping. The fire being stretched out was because of the helium atoms jumping around and releasing into all different directions.
UPDATE:
Oxygen got it's placement in the Periodic Table of the Elements because of its reactivity. This was how the Periodic Table of the Elements was made a long time ago. Electrons rule an elements reactivity. Oxygen has eight protons and eight electrons. It is on the hunt for two more electrons to fill the outer shell and make a stable atom.
There are three oxygen atoms bound to a central nitrogen atom. There is an initial spark of heat that breaks these bonds causing the oxygen to rush away from the Nitrogen to bond with the atoms that it prefers like carbon, hydrogen and even other oxygen atoms leaving the nitrogen to pair up with each other. Every time atoms form a new bond, the reaction releases a new energy which causes the explosion.
Another example of this is in a candle. The carbon atoms in the wick bond with the oxygen atom in the air. Or a very slow process of rust: when iron and oxygen combine.
Light Hypothesis: What is Light?
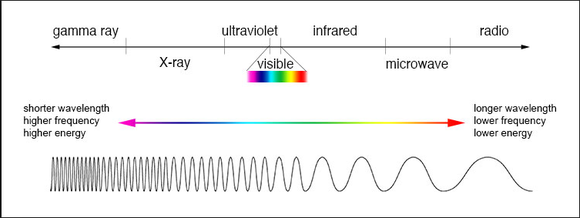
Light is energy. All energy has a different wavelength. These wavelengths are shown on the electromagnetic spectrum, listed from the longest wavelengths to the shortest. Starting with radio waves. These waves are very long and can pass through things easily which allows them to reach farther distances and have no harm to the human body. Although, gamma rays have the shortest wavelength and the highest frequency. Gamma rays can be extremely harmful to your body and even re-order your DNA, completely messing you up. In the middle, there is the visible wavelengths, this is light. This is otherwise known as the rainbow. ROYGBIV: we see these colors through their wavelengths and how intense those wavelengths are. We can only see red, orange, yellow, green, blue, indigo, and violet. In class we burned different chemicals and they all produced different colors. We later learned that because of the different atoms in the chemicals. The way/speed that atoms release energy create different colors. How we see these colors are in wave lengths.
What are the Phases of matter? (Dry Ice Blog Post)
solid to liquid= melting
liquid to solid= freezing
liquid to gas= evaporation
gas to liquid= condensation
solid to gas= sublimation
gas to solid= deposition
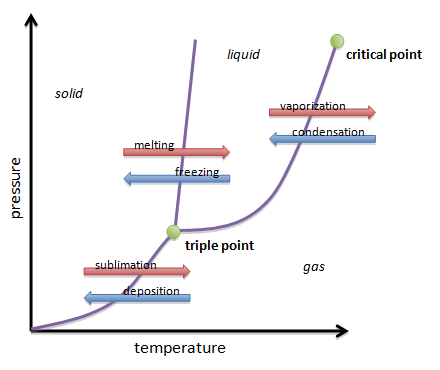
Everything I just explained is shown on the phase change diagram. The phase change diagram shows how one phase of matter can transform change into another. It also shows how temperature affects a phase of matter and how pressure can affect another phase of matter.
The phase change diagram shows that solids and liquids have higher pressures which means those phases or more dense than gas.
When a solid gets hotter, it turns into a liquid. When a liquid gets colder, it turns into a solid.
When a solid gets hotter, it turns into a gas. When a gas gets colder, it turns into a solid. When liquid gets hotter, it turns into a gas. When a gas gets colder, it turns into a liquid.The red arrows means increasing temperature and the blue arrows means decreasing temperature.
Phase change is the what happens when one matter transforms into another. The three phases of matter are solid, liquid, and gas. The only way to change the phase of matter is through temperature and pressure.
liquid to solid= freezing
liquid to gas= evaporation
gas to liquid= condensation
solid to gas= sublimation
gas to solid= deposition
Everything I just explained is shown on the phase change diagram. The phase change diagram shows how one phase of matter can transform change into another. It also shows how temperature affects a phase of matter and how pressure can affect another phase of matter.
The phase change diagram shows that solids and liquids have higher pressures which means those phases or more dense than gas.
When a solid gets hotter, it turns into a liquid. When a liquid gets colder, it turns into a solid.
When a solid gets hotter, it turns into a gas. When a gas gets colder, it turns into a solid. When liquid gets hotter, it turns into a gas. When a gas gets colder, it turns into a liquid.The red arrows means increasing temperature and the blue arrows means decreasing temperature.
Phase change is the what happens when one matter transforms into another. The three phases of matter are solid, liquid, and gas. The only way to change the phase of matter is through temperature and pressure.
Properties of Matter Blog Post
Phases of matter is dictated by temperature. Higher energy = Faster moving molecules
Lower energy= Slower moving molecules
Pressure is the sum of the force of the molecules bouncing off of each other.
Temperature affects pressure, pressure changes volume.
Density=Mass/Volume
Increase volume, density goes down, decrease mass, density goes down.
Floating candle, warm up in order to float.
Negative, neutral & positive buoyancy.
In class we made Floating Lanterns. The plastic bag created a container for the molecules to expand in. When we lit the candles underneath the bag, the oxygen from the flame released into the bag. The temperature heating up the molecules inside of the bag caused them to get bigger and bigger. That makes the mass bigger, but causes the weight of the molecules to burn off. Essentially, it grows in size, but becomes less heavy.
Phases Of Matter Pt. 2:
I was absent all week. I don't know what this is...
Instead of the making rocks lab, I am going to do a bismuth crystallization lab.
I did some initial research on this:
Bismuth crystals are made by melting bismuth and then letting it cool down again. Crystallization will take place and the crystals will start growing. Depening on the volume you use for the molten bismuth, you can either empty it, removing the uncrystallized bismuth (still liquid) or you can take the crystals out of the melt with a pair of tweezers. The crystals have a tendency to float, because the density of liquid bismuth is higher than that of solid bismuth.
The colour of the crystals is caused by a thin layer of bismuth oxide, that is formed when bismuth comes in contact with oxygen at the right temperature. The colour can be any colour from the rainbow, but in general some colours are more abundant than others. There are crystals that are mostly composed of blue and purple, or blauw and green with some yellow.
Wikipedia Information:
Bismuth is a chemical element with the symbol Bi and the atomic number 83. Bismuth, a pentavalent post-transition metal and one of the pnictogens, chemically resembles its lighter homologs arsenic and antimony. Elemental bismuth may occur naturally, although its sulfide and oxide form important commercial ores. The free element is 86% as dense as lead. It is a brittle metal with a silvery white color when freshly produced but is often seen in air with a pink tinge owing to surface oxidation. Bismuth is the most naturally diamagnetic element, and has one of the lowest values of thermal conductivity among metals.
Bismuth is a brittle metal with a white, silver-pink hue, often occurring in its native form, with an iridescent oxidetarnish showing many colors from yellow to blue. The spiral, stair-stepped structure of bismuth crystals is the result of a higher growth rate around the outside edges than on the inside edges. The variations in the thickness of the oxide layer that forms on the surface of the crystal causes different wavelengths of light to interfere upon reflection, thus displaying a rainbow of colors. When burned in oxygen, bismuth burns with a blue flame and its oxide forms yellow fumes.[13] Its toxicity is much lower than that of its neighbors in the periodic table, such as lead, antimony, and polonium.
Instead of the making rocks lab, I am going to do a bismuth crystallization lab.
I did some initial research on this:
Bismuth crystals are made by melting bismuth and then letting it cool down again. Crystallization will take place and the crystals will start growing. Depening on the volume you use for the molten bismuth, you can either empty it, removing the uncrystallized bismuth (still liquid) or you can take the crystals out of the melt with a pair of tweezers. The crystals have a tendency to float, because the density of liquid bismuth is higher than that of solid bismuth.
The colour of the crystals is caused by a thin layer of bismuth oxide, that is formed when bismuth comes in contact with oxygen at the right temperature. The colour can be any colour from the rainbow, but in general some colours are more abundant than others. There are crystals that are mostly composed of blue and purple, or blauw and green with some yellow.
Wikipedia Information:
Bismuth is a chemical element with the symbol Bi and the atomic number 83. Bismuth, a pentavalent post-transition metal and one of the pnictogens, chemically resembles its lighter homologs arsenic and antimony. Elemental bismuth may occur naturally, although its sulfide and oxide form important commercial ores. The free element is 86% as dense as lead. It is a brittle metal with a silvery white color when freshly produced but is often seen in air with a pink tinge owing to surface oxidation. Bismuth is the most naturally diamagnetic element, and has one of the lowest values of thermal conductivity among metals.
Bismuth is a brittle metal with a white, silver-pink hue, often occurring in its native form, with an iridescent oxidetarnish showing many colors from yellow to blue. The spiral, stair-stepped structure of bismuth crystals is the result of a higher growth rate around the outside edges than on the inside edges. The variations in the thickness of the oxide layer that forms on the surface of the crystal causes different wavelengths of light to interfere upon reflection, thus displaying a rainbow of colors. When burned in oxygen, bismuth burns with a blue flame and its oxide forms yellow fumes.[13] Its toxicity is much lower than that of its neighbors in the periodic table, such as lead, antimony, and polonium.
Ocean Acidification
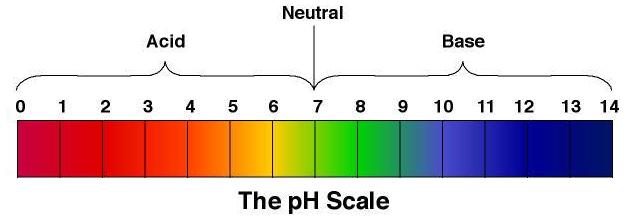
Ocean acidification is when carbon from the atmosphere goes into the ocean causing the Ph levels to rise therefore increasing the acidity of the water. Ph is a way to test acidity in water. Ph measures acidity or basicity. pH is based on the potential amount of hydrogen ions. 30-40% of CO2 produced by humans is dissolved into our oceans. The CO2 dissolved into the water reacts with H20(the water) to form carbonic acid(H2CO3).Carbonic Acid releases one Hydrogen ion, which creates bicarbonate. The bicarbonate can break down even more, to a carbonate ion. The extra Hydrogens lost from the bicarbonate and carbonate ions, cause the pH levels to rise making the ocean more acidic. This is a Ph level scale:
This acidity is bad for the environment because it affects sea life. And the ocean provides for our population and is a main resource for human diet. If the acidity is affecting things at the bottom of the food chain, it also can affect us at the top of the food chain because small fish eat those plants and phytoplankton that absorb the Hydrogen ions and then bigger fish eat those smaller fish, then we eat those larger fish. Calcium carbonate is vital to the health of crustaceans and many other marine animals. Calcium carbonate is made with carbonate ions. The problem here is that the carbonate ions are being joined by the hydrogen ions, creating more bicarbonate and reducing the amount of calcium carbonate in the ocean. Ocean acidification is killing coral and all the other marine life that the ocean has to offer. We don't want to see these things dying so that future generations do not get the opportunity to see and experience them.
Disapearing Ink
Ocean acidification happens when carbon dioxide gets into the ocean and creates a reaction that makes the ocean more acidic. This happens every time carbon dioxide goes into water. We made disappearing ink from phenolphthalein, ethanol, distilled water, and NaOH causing a reaction of carbon dioxide in salt water(sea water).
These were the instructions to the Disappearing Ink lab:
The way that disapearing ink works is that you start out with pure alcohol and salt in water(mimicing sea water), and when the solution is exposed to the air-carbon dioxide, it cancels out the chemicals and just turns into H2O making it become clear, just how water appears. The phenolphthalein is what PH tablets are made of. When you mix the three ingredients together, the mixture is a creamy white. When the sodium hydroxide is dripped in, the compound turns red, this is an example of ocean acidification.
These were the instructions to the Disappearing Ink lab:
- Measure 1g Phenolphthalein into a 125ml Erlenmeyer flask
- Rinse the flask with distilled water
- Fill pipets with 10 milemeter of Ethanol
- Add 100ml D1 H20
- Add (dropwise) 3m Naoh until dark red.
The way that disapearing ink works is that you start out with pure alcohol and salt in water(mimicing sea water), and when the solution is exposed to the air-carbon dioxide, it cancels out the chemicals and just turns into H2O making it become clear, just how water appears. The phenolphthalein is what PH tablets are made of. When you mix the three ingredients together, the mixture is a creamy white. When the sodium hydroxide is dripped in, the compound turns red, this is an example of ocean acidification.